Abstract
Cytogenetic analysis is still an important and mandatory component of Acute Myeloid Leukemia (AML) diagnosis and prognosis. Pretreatment cytogenetic and molecular genetic findings are one of the major independent prognostic markers in AML, and they determine chemotherapy response and outcome. However, cytogenetic does not provide alternative treatments when a patient have a high cytogenetic risk, and requires relatively long time until obtaining the results despite the treatment of these patients should begin as soon as possible.
The aim of this study is providing data about the utility of a new AML Precision Medicine (PM) Test as a complementary tool to conventional cytogenetic to overcome the main obstacles this later has.
For this purpose, AML bone marrow from 111 patients were received at the laboratory 24h from extraction and incubated for 48h in 96-well plates containing single drugs or combinations, representing up to 31 different treatments that are currently given in the clinical practice. The analyses were performed in the automated flow cytometry PharmaFlow platform and the test results can be sent to the hematologists 72h after the extraction of the sample. Pharmacological responses were calculated using pharmacokinetic population models. Induction response was assessed according to the Cheson criteria (2003). Patients attaining a complete remission (CR) or CR with incomplete blood count recovery (CRi) were classified as responders and the remaining as resistant, excluding early deaths. The probability of being resistant or non-responder was modeled using binary logistic generalized additive models (GAM) with Cytarabine (CYT) and Idarubicin (IDA) area under the curve (AUC) data and over the cytogenetic risk (favorable/intermediate/adverse). The empirical ROC curves were calculated for the probabilities of being non-responder from each GAM. Final scores and treatments ranking are based on a therapeutic algorithm that integrates ex vivo activity of single drugs, quantified by the AUC and synergism, referred as α parameter, using a surface interaction model. Clinical and cytogenetic risk data of the patients were monitored and collected.
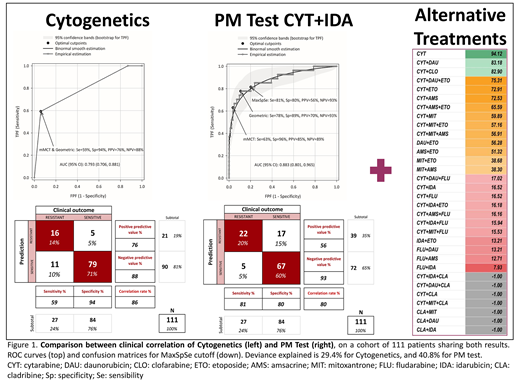
A simple logistic model of the probability of being non-responder over the cytogenetic risk (favorable/intermediate/adverse) explained less variability (29.4%) than the GAM over the AUC values (40.8%) in the subset of 111 patients in whom the cytogenetic risk was informed. Figure 1 shows the results of the clinical correlation of cytogenetics vs PM Test in the cohort of 111 patients analyzed. In both approaches prediction of sensitive patients (Negative Prediction Value, NPV) is better than resistant patients (Positive Predictive Value, PPV), being the PM Test slightly better in predicting the sensitive patients (NPV=93% vs 88%), while the cytogenetics shows a 20% improvement in the prediction of resistant patients (PPV= 76% vs 56% with PM Test). The correlation achieved by the PharmaFlow PM test was 80% that is almost similar than the correlation obtained with the cytogenetic data using the same cut off point (86%). Figure 1 (right) also shows an example of the classification of AML treatments with the PharmaFlow PM Test in a patient sample according to a color scale from higher (green) to lower (red) ex vivo activity.
In summary, despite the PharmaFlow PM Test and cytogenetics provide similar information, results from cytogenetic risk are available typically in 10-14 days, and thus after patient treatment, while results from this novel PM Test are available in 48-72h, prior to treatment. Hence, this novel approach provides information to hematologist with higher predictive value than risk factor (deviance explained 40.8% vs 29.4%) and ahead of treatment, and thus represent a valuable in-time prior to treatment decision making. In addition, the PM Test can provide alternative treatments to AML patient in a basis of their ex vivo activity.
Ballesteros:Vivia Biotech: Employment. Martinez Lopez:Novartis: Research Funding, Speakers Bureau; Celgene: Research Funding, Speakers Bureau; Bristol Myers Squibb: Research Funding, Speakers Bureau; Janssen: Research Funding, Speakers Bureau. Hernandez:Vivia Biotech: Employment. Primo:Vivia Biotech: Employment. Gorrochategui:Vivia Biotech: Employment. Rojas:Vivia Biotech: Employment. Montesinos:Novartis: Research Funding, Speakers Bureau; Daiichi Sankyo: Consultancy, Speakers Bureau.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal